Simultaneous interfacial chemistry and inner Helmholtz plane regulation for superior alkaline hydrogen evolution†
Abstract
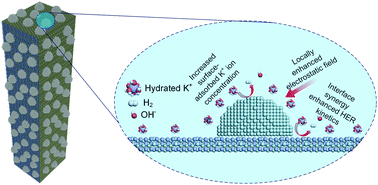
Developing highly efficient and durable alkaline hydrogen evolution reaction (HER) electrocatalysts composed of earth-abundant materials is crucial for large-scale electrochemical hydrogen production. Herein, interfacial chemistry engineering is employed to decouple the restriction of both water discharge and hydrogen adsorption free energy. And low valence state Niδ+ (δ < 1) is proposed as an efficient water-dissociation promoter based on our dual-descriptor method. Moreover, metallic and intrinsic HER-active Ni is introduced and a highly conductive edge-enriched Ni0.2Mo0.8N/Ni hybrid electrocatalyst is constructed. DFT calculations and microkinetics analysis demonstrate that the Ni site and Ni0.2Mo0.8N site have favorable hydroxyl and hydrogen species adsorption energetics, respectively, which can cooperate synergistically towards alkaline hydrogen evolution. The resulting electrocatalyst shows excellent HER electrocatalytic performance with an overpotential of about 70 mV at 300 mA cm−2 and a Tafel slope of 33 mV dec−1. It also offers outstanding operational stability at large current densities up to 200 mA cm−2. Further theoretical calculations suggest a tip-enhanced-like local electric field around the topmost Ni nanoparticles, leading to increased K-ion concentration in the inner Helmholtz plane. The HER kinetics and surface reactant density can be simultaneously improved by this hierarchical Ni0.2Mo0.8N/Ni electrocatalyst. This study might open up new avenues of reasonable design of hierarchical structures for superior electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...