Concentration polarization and metal dendrite initiation in isolated electrolyte microchannels†
Abstract
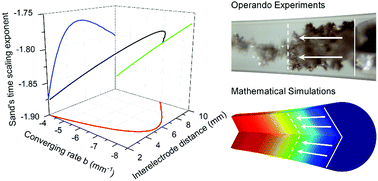
Lithium metal penetrations through the liquid-electrolyte-wetted porous separator and solid electrolytes are a major safety concern of next-generation rechargeable metal batteries. Penetrations were frequently discovered to occur through only a few isolated channels, as revealed by “black spots” on both sides of the separator or electrolyte, which manifest a highly localized ionic flux or current density. Predictions of the penetration time have been difficult due to the hidden and unclear dynamics in these penetration channels. Here, using glass capillary cells, we investigate for the first time the unexpectedly sensitive influence of channel geometry on the concentration polarization and dendrite initiation processes. The characteristic time for the complete depletion of salt concentration at the surface of the advancing electrode, i.e. Sand's time, exhibits a nonlinear dependence on the curvature of the channel walls along the axial direction. While a positively deviated Sand's time scaling exponent can be used to infer a converging penetration area through the electrolyte, a negatively deviated scaling exponent suggests that diffusion limitations can be avoided in expanding channels, such that the fast-advancing tip-growing dendrites will not be initiated. The safety design of rechargeable metal batteries will benefit from considering the true local current densities and the conduction structures.


 Please wait while we load your content...
Please wait while we load your content...