Time-resolved observation of C–C coupling intermediates on Cu electrodes for selective electrochemical CO2 reduction†
Abstract
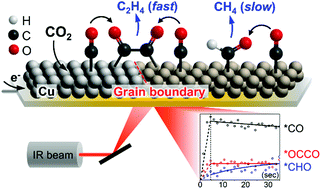
In the electrochemical CO2 reduction reaction (CO2RR), Cu has been spotlighted as the only electro-catalyst that can produce multi-carbon molecules, but the mechanism of the selective C2+ production reaction remains elusive. Here, we directly monitored CO2RR intermediates by employing time-resolved attenuated total reflection-surface enhanced infrared absorption spectroscopy (ATR-SEIRAS), with particular attention to the C1 and C2+ pathways beyond the formation of *CO. Electrodeposited Cu and Cu(OH)2-derived Cu were synthesized, and subsequently employed as a C1 and C2+ activating catalyst and C2+ activating catalyst, respectively. For the first time, a kinetically linked dimer intermediate (*OCCO) was observed and identified as the C2+ path triggering intermediate. The ATR-SEIRAS results suggest that C–C coupling occurs exclusively by CO dimerization toward *OCCO, without the participation of *CHO, which is an intermediate for CH4 production. In the real-time measurements, CO dimerization occurred concurrently with CO adsorption (∼5 s), while proton-coupled reduction toward *CHO has slower kinetics (∼30 s). We demonstrated that the sites showing a high vibrational frequency of *CO on the fragmented Cu surface are the potential active sites for the fast dimerization of CO. This work provides mechanistic insights into the CO2RR pathways and enables the design of efficient C2+-producing catalysts.
- This article is part of the themed collection: 2020 EES Lectureship Winner: Wooyul Kim


 Please wait while we load your content...
Please wait while we load your content...