Suppressing singlet oxygen generation in lithium–oxygen batteries with redox mediators†
Abstract
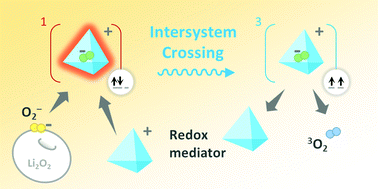
Lithium–oxygen batteries promise high energy but suffer from poor cycle life owing to the generation of highly reactive singlet oxygen 1O2, which rapidly deactivates the electrode and the electrolyte. Here we demonstrate an effective strategy to suppress 1O2 using redox mediators. Using operando spectroscopy techniques, we show that 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO), as a model redox mediator, drastically reduces 1O2 formation, increases the yield of ground state 3O2, and reduces the by-product generation. The 1O2 suppression effect is attributed to an increased intersystem crossing rate induced by redox mediators, with which the relaxation of a singlet-state intermediate to a triplet-state one is accelerated. We further show that this 1O2 suppression effect applies universally across six common redox mediators with up to three orders of magnitude higher 1O2 suppression efficiency compared to 1,4-diazabicyclo[2.2.2]octane (DABCO), the most efficient quencher used in Li–O2 batteries to date. From the comparison of the investigated mediators we discuss the plausible governing principle dictating redox mediators’ effectiveness in suppressing 1O2. Our study achieves effective 1O2 suppression and provides guidelines for designing redox mediators for efficient and reversible lithium–oxygen batteries.


 Please wait while we load your content...
Please wait while we load your content...