In situ structural evolution of the multi-site alloy electrocatalyst to manipulate the intermediate for enhanced water oxidation reaction†
Abstract
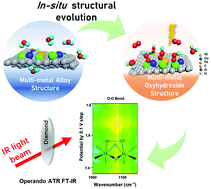
Investigating the reaction mechanism and the rational design of highly efficient electrocatalysts for the oxygen evolution reaction play a key role in renewable energy applications. Here, we report a homogeneous multi-metal-site oxyhydroxide electrocatalyst (consisting of Fe doped NiOOH and Cu doped NiOOH) obtained by in situ electrochemical dealloying of the multi-metal-site alloy (consisting of FeNi3 and NiCu alloys). The in situ structural evolution process manipulates the intermediate and enhances the water oxidation performance. After dealloying, the electrochemically dealloyed catalyst exhibits a small overpotential at large current density (250 mV at 100 mA cm−2), low Tafel slope (34 mV dec−1), remarkably increased ECSA (8-fold larger than before), and superior durability for 200 h at 100 mA cm−2. This electrocatalyst presents one of the best performances among all reported transition metal-based electrocatalysts, and is even superior to the benchmark RuO2. Operando ATR FT-IR reveals that the electrochemically dealloyed electrocatalyst could manipulate the reaction path based on direct O2 evolution mechanism (DOEM) and facilitate the formation of O–O bonds. This fundamental understanding will contribute to the identification and design of the active structure of oxygen evolution electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...