Phosphine vapor-assisted construction of heterostructured Ni2P/NiTe2 catalysts for efficient hydrogen evolution†
Abstract
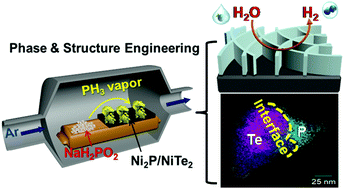
Heterostructured catalysts with unique interfaces and properties endow distinct advantages for many electrochemical reactions. Herein, a phosphine (PH3) vapor-assisted phase and structure engineering strategy is developed for the controllable conversion of non-active NiTe into a heterostructured active Ni2P/NiTe2 catalyst for alkaline hydrogen evolution reaction (HER). The crystalline NiTe2 phase in situ generated in a PH3 vapor environment and the nanosheet morphology both contribute to the outstanding alkaline HER performance with an overpotential of only 62 mV to achieve a current density of −10 mA cm−2. Experimental and DFT mechanistic studies suggest the Ni2P/NiTe2 interfaces provide abundant exposed active sites. The Ni2P/NiTe2 catalyst shows the lowest kinetic barrier for water dissociation and the adsorbed H* can simultaneously bind to two Ni atoms at the interface of Ni2P/NiTe2(011), which greatly enhances the H* binding and HER activities. DFT simulation also shows that more electrons transfer from Ni atoms to H* on Ni2P/NiTe2(011) (0.22 e−) than that on NiTe2(011) (0.13 e−), which explains the enhanced H* binding at the Ni2P/NiTe2(011) interface. The PH3 vapor synthetic approach is also applied to treat other chalcogenide-based materials with low HER activities, such as Ni3S2, to create Ni2P/NiS2 interfaces for significantly enhanced HER activity.


 Please wait while we load your content...
Please wait while we load your content...