Unveiling the mechanisms of lithium dendrite suppression by cationic polymer film induced solid–electrolyte interphase modification†
Abstract
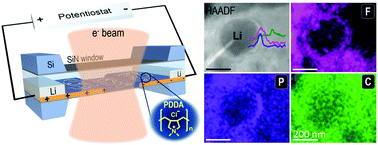
It is crucial to suppress lithium dendrite formation in lithium metal batteries. Formation of a good solid–electrolyte interphase (SEI) has been considered to be effective in limiting lithium dendrite growth. However, how the SEI may be modified during lithium deposition is hard to resolve due to challenges in in situ investigation of the SEI with fine details. We report an in situ study that uncovers the lithium dendrite suppression mechanism arising from SEI modification by a poly(diallyldimethylammonium chloride) (PDDA) cationic polymer film, using electrochemical liquid cell transmission electron microscopy (TEM). Lithium nanogranules are obtained in the presence of the polymer film. Chemical mapping of the deposits provides remarkable details of the SEI on individual nanogranules. It shows that lithium fluorides are uniformly distributed within the inner SEI layer of individual lithium nanogranules, arising from the instantaneous reaction of the deposited lithium with PF6− ions accumulated by the cationic polymer film, and thus the dendritic growth of lithium is prohibited. The ability to directly measure SEI chemistry at the nanoscale down to the individual nanograins in situ and unveil its correlation with the lithium deposition behavior opens future opportunities to explore unsolved mechanisms in batteries.


 Please wait while we load your content...
Please wait while we load your content...