A new lithium diffusion model in layered oxides based on asymmetric but reversible transition metal migration†
Abstract
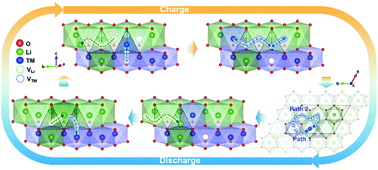
Lithium-rich layered oxides (LLOs) are considered promising cathode materials for lithium-ion batteries because of their high reversible capacity, which is attributed to the exploitation of the novel anionic redox in addition to the conventional cationic redox process. Transition metal (TM) migration, which is known to be the main cause of the voltage decay in LLOs, is now understood to also be the critical factor triggering anionic redox, although this origin is still under debate. A better understanding of the specific TM migration behavior and its effect during charge/discharge would thus enable further development of this class of materials. Herein, we demonstrate that the unique TM migration during charge/discharge significantly alters the lithium diffusion mechanism/kinetics of LLO cathodes. We present clear evidence of the much more sluggish lithium diffusion occurring during discharge (lithiation) than during charge (de-lithiation), which contrasts with the traditional lithium diffusion model based on simple topotactic lithium intercalation/deintercalation in the layered framework. The reversible but asymmetric TM migration in the structure, which originates from the non-equivalent local environments around the TM during the charge and discharge processes, is shown to affect the lithium mobility. This correlation between TM migration and lithium mobility led us to propose a new lithium diffusion model for layered structures and suggests the importance of considering TM migration in designing new LLO cathode materials.


 Please wait while we load your content...
Please wait while we load your content...