Precise anionic regulation of NiFe hydroxysulfide assisted by electrochemical reactions for efficient electrocatalysis†
Abstract
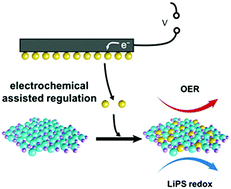
Highly efficient electrocatalysts with high intrinsic activity for oxygen and sulfur redox reactions are strongly required for sustainable energy systems. Generally, cations serve as the real active sites in transition metal compound electrocatalysts, whose electrocatalytic activity is regulated by the surrounding anionic structure. Herein, an electrochemical reaction assisted by an anionic regulation strategy is proposed for precise construction of advanced electrocatalysts with extraordinary electrocatalytic activity. The electrochemical anionic regulation process ensures general release of the regulation reagents for precise substitution of sulfur anions in pristine hydroxide. The as-obtained hydroxysulfide electrocatalyst exhibits a desired electronic structure to afford superb electrocatalytic activity regarding reduced overpotential of 286 mV at 10 mA cm−2 for electrocatalytic oxygen evolution and improved polysulfide redox electrocatalytic activity. This contribution not only renders an emerging strategy for precise regulation of the anionic structure for improved electrocatalytic activity, but also provides information for the rational design of advanced electrocatalysts for sustainable energy applications.
- This article is part of the themed collection: Energy & Environmental Science Cover Art


 Please wait while we load your content...
Please wait while we load your content...