Manipulating the ion-transfer kinetics and interface stability for high-performance zinc metal anodes†
Abstract
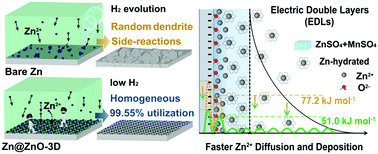
The zinc metal is recognized as one of the most promising anodes for Zn-based batteries in an energy-storage system. However, the deposition and transfer of bivalent Zn2+ into the host structure suffer from sluggish kinetics accompanying the side-reactions at the interface. Herein, we report a new class of Zn anodes modified by a three-dimensional (3D) nanoporous ZnO architecture coating on a Zn plate (designated as Zn@ZnO-3D) prepared by in situ Zn(OH)42− deposition onto the surface. This novel structure has been proven to accelerate the kinetics of Zn2+ transfer and deposition via the electrostatic attraction toward Zn2+ rather than the hydrated one in the electrical double layer. As a consequence, it achieves an average 99.55% Zn utilization and long-time stability for 1000 cycles. Meanwhile, the Zn@ZnO-3D/MnO2 cell shows no capacity fading after 500 cycles at 0.5 A g−1 with a specific capacity of 212.9 mA h g−1. We believe that the mechanistic insight into the kinetics and thermodynamic properties of the Zn metal and the understanding of structure–interface–function relationships are very useful for other metal anodes in aqueous systems.


 Please wait while we load your content...
Please wait while we load your content...