A tailored oxide interface creates dense Pt single-atom catalysts with high catalytic activity†
Abstract
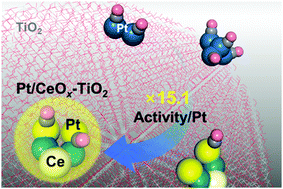
Highly reactive dense Pt single-atoms stabilized on an oxide support can resolve a grand challenge in the economic use of Pt in catalysis. The maximized number density of reaction sites provided by dense Pt single-atoms guarantees the improved catalytic performance of Pt combined with high efficiency. By manipulating the chemical nature of multi-component interfaces, we synthesized CO-tolerant dense Pt single-atoms highly reactive for the CO oxidation reaction, which governs the key steps for chemical energy conversion and emission control. The addition of 1 wt% of Ce to TiO2 support particles creates a CeOx–TiO2 interface that stabilizes Pt single-atoms by strong electronic interactions. Dense Pt single-atoms formed on CeOx/TiO2 oxides exhibit 15.1 times greater specific mass activity toward CO oxidation at 140 °C compared with a bare Pt/TiO2 catalyst. We elaborate how the CeOx–TiO2 interfaces activate the interface-mediated Mars–van Krevelen mechanism of CO oxidation and protect Pt single-atoms from CO-poisoning. Through a comprehensive interpretation of the formation and activation of dense Pt single-atoms using operando X-ray absorption spectroscopy, density functional theory calculations, and experimental catalyst performance tests, we provide a key that enables the catalytic performance of noble metal single-atom catalysts to be optimized by atomic-scale tuning of the metal–support interface.
- This article is part of the themed collection: Energy & Environmental Science Cover Art


 Please wait while we load your content...
Please wait while we load your content...