Heterointerface engineering for enhancing the electrochemical performance of solid oxide cells
Abstract
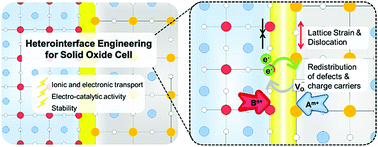
Solid oxide cells (SOCs) have the potential to be the most efficient energy storage and conversion systems. To minimize energy loss due to charge and mass transport associated with the operation of SOC systems at intermediate temperatures, electrodes and electrolytes containing different types of heterointerfaces have been designed, fabricated, and tested under various conditions. While heterointerfaces can significantly enhance not only the ionic and/or electronic conductivity but also the electrocatalytic activity and stability of SOC components, as predicted by theoretical calculations and demonstrated by experimental results, the mechanisms of these enhancements are yet to be fully understood. In this review, we start with an overview of the techniques for fabrication of heterointerfaces with controlled composition, structure, and morphology. Then, the latest developments in performance enhancement of SOCs with heterointerfaces are summarized, including boosting the ionic conductivity of heterostructured electrolytes (oxygen ion conductors and proton conductors) and increasing the electrocatalytic activity and durability of heterostructured electrodes (oxygen electrodes and fuel electrodes). Subsequently, we will highlight the unique attributes of heterointerfaces in the enhancement of the SOC performance and provide important insights into the mechanisms of performance enhancement in order to establish the scientific basis for rational design of better electrolyte and electrode materials. Finally, the remaining challenges in design and fabrication of novel materials for advanced solid-state electrochemical systems will be discussed, together with possible strategies to overcome these critical issues, new research directions, and future perspectives.
- This article is part of the themed collection: Energy & Environmental Science Cover Art


 Please wait while we load your content...
Please wait while we load your content...