The electronic structure of a β-diketiminate manganese hydride dimer†
Abstract
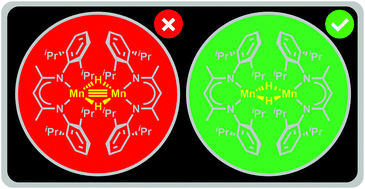
The electronic structure of a dimeric manganese hydride catalyst supported by β-diketiminate ligands, [(2,6-iPr2PhBDI)Mn(μ-H)]2, was investigated with density functional theory. A triple bond between the manganese centres was anticipated from simple electron-counting rules; however, calculations revealed Mn–Mn Mayer bond orders of 0.21 and 0.27 for the ferromagnetically-coupled and antiferromagnetically-coupled extremes, respectively. In accordance with experimentally determined Heisenberg exchange coupling constants of −15 ± 0.1 cm−1 (SQUID) and −10.2 ± 0.7 cm−1 (EPR), the calculated J0 value of −10.9 cm−1 confirmed that the ground state involves antiferromagnetic coupling between high spin Mn(II)-d5 centres. The effect of steric bulk on the bond order was examined via a model study with the least sterically-demanding version of the β-diketiminate ligand and was found to be negligible. Mixing between metal- and β-diketiminate-based orbitals was found to be responsible for the absence of a metal–metal multiple bond. The bridging hydrides give rise to a relatively close positioning of the metal centres, while bridging atoms possessing 2p orbitals result in longer Mn–Mn distances and more stable dimers. The synthesis and characterization of the bridging hydroxide variant, [(2,6-iPr2PhBDI)Mn(μ-OH)]2, provides experimental support for these assessments.


 Please wait while we load your content...
Please wait while we load your content...