Imidazole fused phenanthroline (PIP) ligands for the preparation of multimodal Re(i) and 99mTc(i) probes†
Abstract
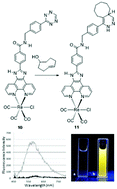
A small library of [2 + 1] 99mTc(I) complexes based on phenyl-imidazole-fused phenanthroline (PIP) ligands were synthesized and evaluated as multimodal molecular imaging probes. Using either a two-step or a one-pot synthesis method, 99mTc-PIP complexes containing N-methylimidazole as the monodentate ligand were prepared and isolated in good (54 to 89%) radiochemical yield, with the exception of one derivative bearing a strongly electron-withdrawing substituent. The stability of the [2 + 1] complexes was assessed in saline and in cysteine and histidine challenge studies, showing 6 hours stability, making them suitable for in vivo studies. In parallel, the Re(I) analogues were prepared as reference standards to verify the structure of the 99mTc complexes. The optical properties were consistent with other previously reported [2 + 1] type Re(I) complexes that have been used as cellular dyes and sensors. To facilitate the development of targeted derivatives, a tetrazine-PIP ligand was also synthesized. The 99mTc complex of the tetrazine PIP ligand effectively coupled to compounds containing a trans-cyclooctene (TCO) group including a TCO-albumin derivative, which was prepared as a model targeting molecule. An added benefit of the Re-PIP-Tz construct is that the emission from the metal complex was quenched by the presence of the tetrazine. Following the addition of TCO, there was a 70-fold increase in fluorescence emission, which can in future be leveraged during in vitro studies to reduce background signal.


 Please wait while we load your content...
Please wait while we load your content...