From lamellar net to bilayered-lamella and to porous pillared-bilayer: reversible crystal-to-crystal transformation, CO2 adsorption, and fluorescence detection of Fe3+, Al3+, Cr3+, MnO4−, and Cr2O72− in water†
Abstract
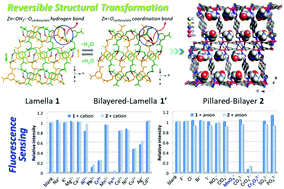
An aqua-coordinated lamellar net [Zn(5-NH2-1,3-bdc)(H2O)] (1, 5-NH2-1,3-H2bdc = 5-amino-1,3-benzenedicarboxylic acid) has been found to undergo a reversible stimuli-responsive 2D-to-2D crystal-to-crystal transformation with a water-free bilayered-lamellar net [Zn(5-NH2-1,3-bdc)] (1′) upon removal and rebinding of aqua ligands, whereas a 2D porous pillared-bilayer [Zn2(5-NH2-1,3-bdc)2(NI-bpy-44)]·DMF (2, NI-bpy-44 = N-(pyridin-4-yl)-4-(pyridin-4-yl)-1,8-naphthalimide) has been tailored by introducing NI-bpy-44 to replace the coordinated aqua ligands. Pillared-bilayer 2 displayed a moderate CO2 uptake of 79.1 cm3 g−1 STP at P/P0 = 1 and 195 K with an isosteric heat of CO2 adsorption (Qst) of 37.0 kJ mol−1 at zero-loading. It is noteworthy that the water suspensions of 1 and 2 both displayed good fluorescence performances, which were effectively quenched by Fe3+, MnO4−, and Cr2O72− ions and shifted to long wavelengths by Fe3+, Al3+, and Cr3+, even with the coexistence of equal amounts of most other interfering ions. Taking the Stern–Volmer quenching constant, limit of detection, quenching efficiency, anti-interference ability, and visual observation into consideration, it is clear that both 1 and 2 are promising and excellent fluorescent sensors for highly sensitive detection of Fe3+, MnO4−, and Cr2O72−.


 Please wait while we load your content...
Please wait while we load your content...