Au54(Et3P)18Cl12: a structurally related cluster to Au32(Et3P)12Cl8 gives insight into the formation process†
Abstract
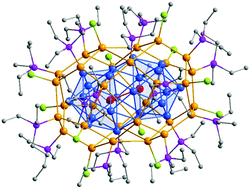
The reaction of Et3PAuCl with NaBH4 in EtOH leads to the metalloid gold cluster Au32(Et3P)12Cl8 (Au32) or Au54(Et3P)18Cl12 (Au54) depending on the work-up procedure of the reaction mixture. The molecular structure of Au54 is determined by X-ray diffraction and can be described as a fusion of two Au32 clusters showing a similar solubility. The metalloid cluster Au54 can be either described by a shell model or as a combination of tetrahedral Au4X units (X = Cl, Et3P); edge and face sharing, whereas tetrahedral Au4 units are a central motif in gold cluster chemistry. This novel Au54 gold cluster gives another unique insight into the formation or decomposition process of metalloid clusters, indicating that Au32 and Au54 form from a single yet unknown cluster source.


 Please wait while we load your content...
Please wait while we load your content...