The synthesis of cyclometalated platinum(ii) complexes with benzoaryl-pyridines as C^N ligands for investigating their photophysical, electrochemical and electroluminescent properties†
Abstract
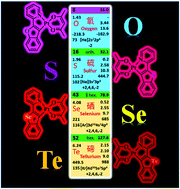
A series of (C^N)Pt(acac)-type complexes has been successfully synthesized with a benzo[b]furan, benzo[b]thiophene, benzo[b]selenophene, or benzo[b]tellurophene group in the benzoaryl-pyridine ligand. Using X-ray crystallography, the chemical structures of the complexes with benzo[b]selenophene and benzo[b]tellurophene groups have been clearly revealed. The photophysical, electrochemical, and electroluminescent (EL) behaviors of these (C^N)Pt(acac)-type complexes have been fully characterized. Furthermore, both time-dependent functional theory (TD-DFT) and natural transition orbital (NTO) theoretical results have been obtained to gain insight into the absorption and emission features. It has been shown that both the absorption bands with the lowest energy and the phosphorescence emission behaviors are dominated by the benzoaryl-pyridine cyclometalating ligand. Importantly, the effects of the group VIA atoms on the properties of these (C^N)Pt(acac)-type complexes have been revealed. Owing to the rareness of (C^N)Pt(acac)-type complexes with benzo[b]selenophene and benzo[b]tellurophene groups, their EL abilities have been characterized using solution-processed organic light-emitting diodes (OLEDs). The optimized red OLEDs with the complex bearing a benzo[b]selenophene unit show a maximum external quantum efficiency (ηext) of 6.3%, current efficiency (ηL) of 10.5 cd A−1, and power efficiency (ηP) of 9.1 lm W−1, while the EL device with the complex bearing a benzo[b]tellurophene unit can give deep-red emission at ca. 636 nm with ηext of 6.3%, ηL of 6.5 cd A−1, and ηP of 5.8 lm W−1. This research not only provides novel (C^N)Pt(acac)-type complexes, but also furnishes critical information regarding the photophysical and EL behavior of these new complexes.


 Please wait while we load your content...
Please wait while we load your content...