Induction, control, and rationalization of supramolecular chirogenesis using metalloporphyrin tweezers: a structure-function correlation†
Abstract
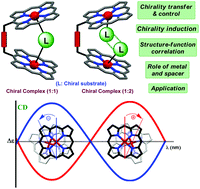
Supramolecular chirogenesis is one of the most rudimentary topics in the interdisciplinary sciences and essentially deals with various natural processes and innovative modern technologies. A comprehensive and rigorous understanding of such phenomenon is necessary to have a clear insight into the fundamental mechanisms and the various controlling factors, which would eventually lead to a range of practical applications of chiral supramolecular science. Metalloporphyrin tweezers have been extensively employed for such chirogenic processes due to their exciting physicochemical and tunable spectral properties, large stabilities, easily available synthetic protocols, and excellent abilities to form molecular assemblies. During the last few decades, various metalloporphyrin tweezers have been developed and considerably utilized by several research groups for assigning the absolute configuration to a variety of chiral diamines, conjugates of primary and secondary amines, amino alcohols, secondary alcohols, α-chiral carboxylic acids, etc. Our group has been at the forefront in trying to establish the structure–property correlation in this important area of interdisciplinary research. A brief account of our systematic investigation for understanding the underpinning mechanism of chirality induction and control at the molecular level over the last few years is presented in this Perspective article. The comprehensive understanding of such mechanistic details will be helpful in understanding various natural processes and designing modern technologies for various chirogenic functions in the fields of molecular sensors, nanotechnology, and supramolecular chemistry.
- This article is part of the themed collection: 2020 Frontier and Perspective articles


 Please wait while we load your content...
Please wait while we load your content...