Luminescent halogen-substituted 2-(N-arylimino)pyrrolyl boron complexes: the internal heavy-atom effect†
Abstract
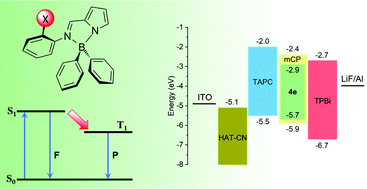
A group of new boron complexes [BPh2{κ2N,N′-NC4H3-2-C(H)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N-C6H4X}] (X = 4-Cl 4c, 4-Br 4d, 4-I 4e, 3-Br 4f, 2-Br 4g, 2-I 4h) containing different halogens as substituents in the N-aryl ring have been synthesized and characterized in terms of their molecular properties. Their photophysical characteristics have been thoroughly studied in order to understand whether these complexes exhibit an internal heavy-atom effect. Phosphorescence emission was found for some of the synthesized halogen-substituted boron molecules, particularly for 4g and 4h. DFT and TDDFT calculations showed that the lower energy absorption band resulted from the HOMO to LUMO (π–π*) transition, except for 2-I 4h, where the HOMO−1 to LUMO transition was also involved. The strong participation of iodine orbitals in HOMO−1 is reflected in the calculated absorption spectra of the iodine derivatives, especially 2-I 4h, when spin–orbit coupling (SOC) was included. Organic light-emitting diodes (OLEDs) based on these complexes, in the neat form or dispersed in a matrix, were also fabricated and tested. The devices based on films prepared by thermal vacuum deposition showed the best performance. When neat complexes were used, a maximum luminance (Lmax) of 1812 cd m−2 was obtained, with a maximum external quantum efficiency (EQEmax) of 0.15%. An EQEmax of ca. 1% along with a maximum luminance of 494 cd m−2 were obtained for a device fabricated by co-deposition of the boron complex and a host compound (1,3-bis(N-carbazolyl)benzene, mCP).
N-C6H4X}] (X = 4-Cl 4c, 4-Br 4d, 4-I 4e, 3-Br 4f, 2-Br 4g, 2-I 4h) containing different halogens as substituents in the N-aryl ring have been synthesized and characterized in terms of their molecular properties. Their photophysical characteristics have been thoroughly studied in order to understand whether these complexes exhibit an internal heavy-atom effect. Phosphorescence emission was found for some of the synthesized halogen-substituted boron molecules, particularly for 4g and 4h. DFT and TDDFT calculations showed that the lower energy absorption band resulted from the HOMO to LUMO (π–π*) transition, except for 2-I 4h, where the HOMO−1 to LUMO transition was also involved. The strong participation of iodine orbitals in HOMO−1 is reflected in the calculated absorption spectra of the iodine derivatives, especially 2-I 4h, when spin–orbit coupling (SOC) was included. Organic light-emitting diodes (OLEDs) based on these complexes, in the neat form or dispersed in a matrix, were also fabricated and tested. The devices based on films prepared by thermal vacuum deposition showed the best performance. When neat complexes were used, a maximum luminance (Lmax) of 1812 cd m−2 was obtained, with a maximum external quantum efficiency (EQEmax) of 0.15%. An EQEmax of ca. 1% along with a maximum luminance of 494 cd m−2 were obtained for a device fabricated by co-deposition of the boron complex and a host compound (1,3-bis(N-carbazolyl)benzene, mCP).


 Please wait while we load your content...
Please wait while we load your content...