Oxidative addition of tetrathiocins to palladium(0) and platinum(0): a route to dithiolate coordination complexes†
Abstract
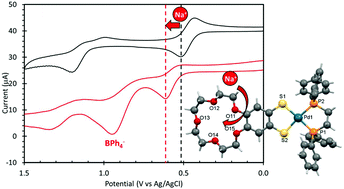
The preparation of a series of 4,4′,5,5′-substituted benzo-fused 1,2,5,6-tetrathiocins X2C6H2S4C6H2X2 (1a–1g) were prepared from the reaction of S2Cl2 with 1,2-C6H4X2 (X = OMe, OEt; X2 = OCH2O, OCH2CH2O, OCH2CH2CH2O, MeNC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)NMe, O(CH2CH2O)4). The oxidative addition of 1a–1g to zero-valent Pd2dba3 or Pt2dba3 (dba = dibenzylideneacetone) in the presence of bis (diphenylphosphino)ethane (dppe) resulted in formation of the substituted mononuclear benzenedithiolate complexes M(L)(dppe) [L = dithiolate ligand; 2a–2g (M = Pd) and 3a–3g (M = Pt)] in 37–89% yield based on recrystallized material. Representative examples of M(L)(dppf) [dppf = bis-diphenylphosphinoferrocene; 4a, 4g (M = Pd) and 5g (M = Pt)] were prepared in a similar fashion. The structures of all derivatives were determined by X-ray diffraction, multinuclear NMR and elemental analysis. The reactivity of the two crown ether dithiolate complexes, 2g and 4g, with 1 equivalent of NaBPh4 led to isolation of the 1 : 1 complexes in which the Na+ cation is bound in the macrocyclic crown, [Na(2g)(MeOH)2][BPh4] and [Na(4g)][BPh4] whose structures were determined by X-ray diffraction. Electrochemical studies supported through DFT calculations on the crown ether derivatives revealed a series of ligand-based oxidation waves corresponding to the dithiolate ligand (and dppf for 4g and 5g) whose redox potentials were shifted by ca. +0.1 V on binding to Na+.
O)NMe, O(CH2CH2O)4). The oxidative addition of 1a–1g to zero-valent Pd2dba3 or Pt2dba3 (dba = dibenzylideneacetone) in the presence of bis (diphenylphosphino)ethane (dppe) resulted in formation of the substituted mononuclear benzenedithiolate complexes M(L)(dppe) [L = dithiolate ligand; 2a–2g (M = Pd) and 3a–3g (M = Pt)] in 37–89% yield based on recrystallized material. Representative examples of M(L)(dppf) [dppf = bis-diphenylphosphinoferrocene; 4a, 4g (M = Pd) and 5g (M = Pt)] were prepared in a similar fashion. The structures of all derivatives were determined by X-ray diffraction, multinuclear NMR and elemental analysis. The reactivity of the two crown ether dithiolate complexes, 2g and 4g, with 1 equivalent of NaBPh4 led to isolation of the 1 : 1 complexes in which the Na+ cation is bound in the macrocyclic crown, [Na(2g)(MeOH)2][BPh4] and [Na(4g)][BPh4] whose structures were determined by X-ray diffraction. Electrochemical studies supported through DFT calculations on the crown ether derivatives revealed a series of ligand-based oxidation waves corresponding to the dithiolate ligand (and dppf for 4g and 5g) whose redox potentials were shifted by ca. +0.1 V on binding to Na+.


 Please wait while we load your content...
Please wait while we load your content...