Monitoring inorganic pyrophosphatase activity with the fluorescent dizinc(ii) complex of a macrocycle bearing one dansylamidoethyl antenna†
Abstract
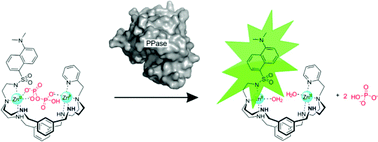
The dizinc(II) complexes of L were used for the recognition of anions by fluorescence spectroscopy (L is a heteroditopic hexaazamacrocycle with two diethylenetriamine coordination heads with 2-methylpyridyl and dansylamido ethyl arms, and m-xylyl spacers). The protonation of L and stability constants of its zinc(II) complexes were determined in aqueous solution, at 298.2 ± 0.1 K and I = 0.10 ± 0.01 M in KNO3. At a 2 : 1 Zn2+/L ratio, the dinuclear complexes clearly dominate. The ligand alone does not display fluorescence changes upon increasing the pH value, but in the presence of Zn2+ the emission reaches a maximum at pH ≅ 7.5, at which 95% of the ligand is in the dinuclear complex form. The emission appears concomitantly with the [Zn2H−1L]3+ species formation, which supports that the latter complex corresponds to the metal-promoted deprotonation of dansylamide NH. The [Zn2H−1L]3+ complexes were used for the recognition of phosphate and polyphosphate anions in aqueous solution buffered at pH 7.5 with 2 mM PIPPS, at 298.2 K. The binding of anions causes a decrease of the emission. The association constant determination revealed that HPPi3− is the strongest bound anion (log Kapp = 5.57), followed by HATP3− (two times weaker), and the remaining anions show lower binding constants, with HPO42− having the weakest uptake by the receptor. The observed selectivity of the [Zn2H−1L]3+ receptor for PPi in relation to HPO42−, and the fact that the formation of the [Zn2H−1L]3+ complex is not disturbed by the presence of Mg2+, allowed monitoring of the PPi hydrolysis by using inorganic pyrophosphatase in real-time.


 Please wait while we load your content...
Please wait while we load your content...