IrCo alloy nanoparticles supported on N-doped carbon for hydrogen evolution electrocatalysis in acidic and alkaline electrolytes†
Abstract
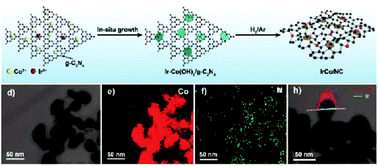
Designing efficient and low-cost electrocatalysts for the hydrogen evolution reaction (HER) is of great importance to advance water splitting technology towards practical applications. Herein, we report the preparation of IrCo nanoparticles supported on nitrogen-doped carbon (IrCo/NC) as a HER electrocatalyst in acidic and alkaline electrolytes. The IrCo/NC composite is obtained by pyrolyzing an Ir-doped Co(OH)2 precursor on g-C3N4, and is endowed with N-doped carbon and uniform IrCo alloy nanoparticles via a crystal confinement resulting from the Ir-doping into the Co(OH)2 layer. Electrocatalytic analysis shows that the IrCo/NC electrode requires low overpotentials of 32 mV at 10 mA cm−2 in 0.5 M H2SO4 and 33 mV in 1 M KOH, which are superior to those of the Co/NC and IrCo alloys that are free of Ir-doping or N-doped carbon. The results provide a strategy for designing and preparing active noble–transition bimetallic alloy electrocatalysts as efficient HER catalysts.


 Please wait while we load your content...
Please wait while we load your content...