Luminescent Zn(ii) and Cd(ii) complexes with chiral 2,2′-bipyridine ligands bearing natural monoterpene groups: synthesis, speciation in solution and photophysics†
Abstract
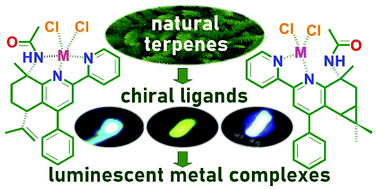
Mononuclear zinc(II) and cadmium(II) complexes, ZnLCl2 (1), CdLCl2 (2), ZnL1Cl2·2H2O (3), and CdL1Cl2·2H2O (4), with chiral ligands containing a 2,2′-bipyridine moiety and natural terpene (+)-limonene (L) or (+)-3-carene (L1) moieties were synthesized. In these complexes the L and L1 ligands are shown to coordinate Zn2+ and Cd2+ ions through the 2,2′-bipyridine moiety. The acetamide group of the ligands interacts with M2+ ions by forming N⋯M2+ and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯M2+ contacts and N–H⋯Cl hydrogen bonds with coordinated Cl− ions. In solutions the complexes have several conformers differing by the degree of the turn of the acetamide moiety relative to the ligand core and the type of its interaction with the coordination core. The ligands and complexes exhibit luminescence with the quantum yield increasing in the order: ligand < cadmium(II) complex < zinc(II) complex. The complexes 3 and 4 demostrate excitation wavelength independent single-channel fluorescence. As opposed to 3 and 4, the complexes 1 and 2 demonstrate excitation wavelength dependent emission with nanosecond and microsecond lifetimes of the excited states. According to our TD-DFT calculations, an interplay of ligand centered and halide to ligand transitions facilitates two deactivation channels in 1 and 2: S1–S0 and T1–S0.
O⋯M2+ contacts and N–H⋯Cl hydrogen bonds with coordinated Cl− ions. In solutions the complexes have several conformers differing by the degree of the turn of the acetamide moiety relative to the ligand core and the type of its interaction with the coordination core. The ligands and complexes exhibit luminescence with the quantum yield increasing in the order: ligand < cadmium(II) complex < zinc(II) complex. The complexes 3 and 4 demostrate excitation wavelength independent single-channel fluorescence. As opposed to 3 and 4, the complexes 1 and 2 demonstrate excitation wavelength dependent emission with nanosecond and microsecond lifetimes of the excited states. According to our TD-DFT calculations, an interplay of ligand centered and halide to ligand transitions facilitates two deactivation channels in 1 and 2: S1–S0 and T1–S0.


 Please wait while we load your content...
Please wait while we load your content...