Photocatalytic, dye degradation, and bactericidal behavior of Cu-doped ZnO nanorods and their molecular docking analysis
Abstract
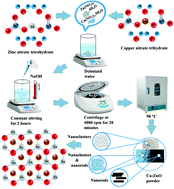
Nanostructures of Cu-doped ZnO (Cu:ZnO) were prepared with the chemical precipitation technique with an aim to enhance the photocatalytic and antibacterial properties of ZnO. Phase constitution, the presence of functional groups, optical properties, elemental composition, surface morphology and microstructure were evaluated using an X-ray diffractometer (XRD), Fourier transform infrared spectroscopy (FTIR), UV-Vis spectrophotometer, energy dispersive X-ray spectroscopy (EDS), field emission scanning electron microscope (FESEM) and high resolution transmission electron microscope (HR-TEM), respectively. Emission spectra were obtained with a photoluminescence (PL) spectroscope whereas interlayer d-spacing was estimated through HR-TEM. ZnO consisted of a hexagonal wurtzite structure. The crystallinity of the sample was observed to increase with increasing doping concentration. The addition of Cu to ZnO served to transform nanoclusters into nanorods as revealed during SEM analysis. Catalytic activity enhanced due to the formation of nanorods, and UV-Vis absorption spectra showed that methylene blue (MB) degraded more efficiently with ZnO nanoclusters compared to the NaBH4 reagent. In addition, the doped NPs showed enhanced bacterial efficiency for G +ve. Finally, a molecular docking study was undertaken to highlight the importance of the binding interactions of the Cu-doped ZnO nanorods with β-lactamase and beta-ketoacyl-acyl carrier protein synthase III (FabH) as possible enzyme targets. This research indicates that Cu-doped Zn nanorods are a highly efficient photocatalyst and can be aptly employed for wastewater treatment and antibacterial applications.


 Please wait while we load your content...
Please wait while we load your content...