Influence of Eu valence on the optical activity of BaTiO3 decorated with CaF2 synthesized by microwave-assisted hydrothermal method
Abstract
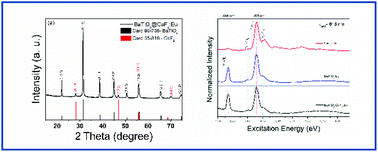
Inorganic hybrid materials have promising applications in absorbers and for the photon harvesting of solar irradiation, such as in DSSC photoanodes. Moreover, investigation of the interactions between the photoanode constituent materials is extremely important, since it is known that the properties of the materials are strongly dependent on the nucleation and growth process. Therefore, the purpose of this work was to synthesize a system consisting of a synergic combination of two inorganic hosts, BaTiO3 and CaF2, synthesized together through a microwave-assisted hydrothermal method, which allows single-phase materials to be obtained after short synthesis times and at low temperatures. The increase in optical activity was investigated after the insertion of Eu ions. From the structural results, it was possible to observe that the method used to obtain the system was efficient, even using temperatures as low as 140 °C and a reaction time of one minute. The optical properties (emission and excitation) were evaluated in the visible and VUV region from 2.5 to 10.5 eV. The XANES Eu LIII edge differences associated with the XEOL results indicate that the synthesis method incorporated Eu3+ in the hosts and the surface of the two compounds, BaTiO3 and CaF2. Therefore, the combination between the Eu3+ activators and the F centers of the host increased the range for photon harvesting, making these systems a promising material for future applications in photoanodes for DSSCs.


 Please wait while we load your content...
Please wait while we load your content...