Abnormal co-doping effect on the red persistent luminescence SrS:Eu2+,RE3+ materials†
Abstract
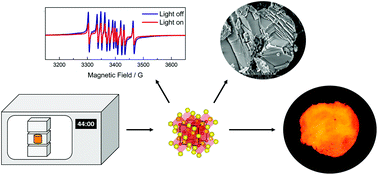
Persistent luminescence materials are a reality in several applications. However, there is still a lack of efficient red-emitting materials. SrS:Eu2+ phosphor is a potential candidate since its strong nephelauxetic effect shifts Eu2+4f65d1 → 4f7 to red, and its weak bond between strontium and sulphide, due to the soft base-hard acid character, generates a high number of intrinsic defects. SrS:Eu2+,RE3+ materials were efficiently prepared by two rounds of 22 min microwave-assisted solid-state synthesis. The highly crystalline purity and the material organization at the micro-scale were observed with X-ray powder diffraction and scanning electron microscopy, respectively. X-ray absorption spectroscopy revealed a low amount of Eu2+ compared to Eu3+ due to the efficient Eu2+ photo-oxidation by X-ray irradiation in the high storage capability SrS host matrix. Electron paramagnetic resonance spectra confirmed that at least 50% of Eu2+ ions in the material are photo-oxidized during excitation, reinforcing the previously established mechanisms. The RE2+ energy level positioned very close to or into the conduction band led to an abnormal co-doping effect, with similar effects independent of the co-dopant. The high concentration of intrinsic defects in SrS indicates that the soft–hard pair host is an excellent approach to develop efficient persistent luminescence materials.
- This article is part of the themed collection: New Talent: Americas


 Please wait while we load your content...
Please wait while we load your content...