Stabilization of a mixed iron vanadium based hexagonal tungsten bronze hydroxyfluoride HTB–(Fe0.55V0.45)F2.67(OH)0.33 as a positive electrode for lithium-ion batteries†
Abstract
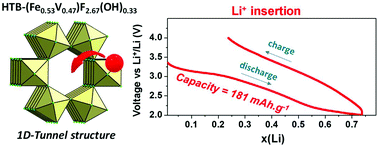
In our search for novel insertion compounds for Li-based batteries, we have identified a new mixed iron vanadium based Hexagonal Tungsten Bronze (HTB) type phase. Its synthesis involves two steps which consist first of preparing mixed metal hydrated fluoride Fe1.64V1.36F8(H2O)2 by a microwave assisted thermal process, followed by thermal treatment under air to obtain metastable HTB–(Fe0.55V0.45)F2.67(OH)0.33 hydroxyfluoride. 57Fe Mössbauer spectrometry demonstrates the presence of oxidation states Fe2+ and Fe3+ in Fe1.64V1.36F8(H2O)2 as opposed to only Fe3+ in HTB–(Fe0.55V0.47)F2.67(OH)0.33. Moreover, the Mössbauer spectra recorded at 77 K reveal that none of the compounds shows magnetic ordering owing to the presence of V3+ distributed over the crystallographic sites of Fe3+. Complementary X-ray spectroscopy and Rietveld refinement further confirm the successful synthesis of HTB–(Fe0.55V0.45)F2.67(OH)0.33. Electrochemically, the new HTB–(Fe0.55V0.45)F2.67(OH)0.33 shows a first discharge capacity of 181 mA h g−1 with 67% of this capacity remaining upon cycling. Unlike HTB–FeF2.66(OH)0.34, the structure remains stable after the first discharge confirming the positive effect of vanadium in the HTB network.


 Please wait while we load your content...
Please wait while we load your content...