Amorphous co-doped MoSex effectively enhances photocatalysis in visible light
Abstract
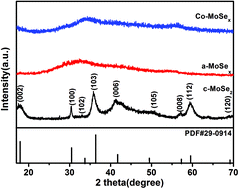
Molybdenum selenide has great potential for the degradation of water pollutants, but its performance is limited by the active sites and recombination of photogenerated electron–hole pairs. In this study, an amorphous co-doped MoSex (Co-MoSex) sample was synthesized by a low-temperature hydrothermal method. Three types of unsaturated Se–Se bonds were found in Co-MoSex by Raman spectroscopy: bridging diselenide, shared diselenide, and terminal diselenide. Photocatalytic experimental results revealed that the degradation rate of Co-MoSex to methylene blue was 96.3%, the degradation rate of amorphous MoSex (a-MoSex) was 61.6%, and the degradation rate of crystalline molybdenum selenide (c-MoSe2) was 23.2%. Furthermore, it was found that the band gap of the Co-MoSex photocatalyst was reduced by 0.15 eV compared to the a-MoSex photocatalyst. The photocatalytic activity of the co-doped MoSex was enhanced by increasing the unsaturated Se atom active sites, reducing the recombination of photogenerated electron–hole pairs, and improving light utilization.


 Please wait while we load your content...
Please wait while we load your content...