The role of the capping agent and nanocrystal size in photoinduced hydrogen evolution using CdTe/CdS quantum dot sensitizers†
Abstract
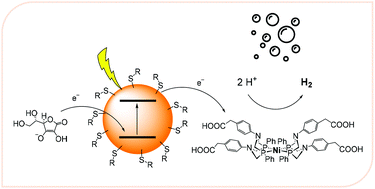
Hydrogen production via light-driven water splitting is a key process in the context of solar energy conversion. In this respect, the choice of suitable light-harvesting units appears as a major challenge, particularly as far as stability issues are concerned. In this work, we report on the use of CdTe/CdS QDs as photosensitizers for light-assisted hydrogen evolution in combination with a nickel bis(diphosphine) catalyst (1) and ascorbate as the sacrificial electron donor. QDs of different sizes (1.7–3.4 nm) and with different capping agents (MPA, MAA, and MSA) have been prepared and their performance assessed in the above-mentioned photocatalytic reaction. Detailed photophysical studies have been also accomplished to highlight the charge transfer processes relevant to the photocatalytic reaction. Hydrogen evolution is observed with remarkable efficiencies when compared to common coordination compounds like Ru(bpy)32+ (where bpy = 2,2′-bipyridine) as light-harvesting units. Furthermore, the hydrogen evolution performance under irradiation is strongly determined by the nature of the capping agent and the QD size and can be related to the concentration of the surface defects within the semiconducting nanocrystal. Overall, the present results outline how QDs featuring large quantum yields and long lifetimes are desirable to achieve sustained hydrogen evolution upon irradiation and that a precise control of the structural and photophysical properties thus appears as a major requirement towards profitable photocatalytic applications.


 Please wait while we load your content...
Please wait while we load your content...