Ru(ii)-Naphthoquinone complexes with high selectivity for triple-negative breast cancer†
Abstract
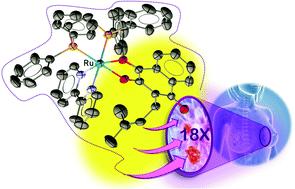
Six new ruthenium(II) complexes with lapachol (Lap) and lawsone (Law) with the general formula [Ru(L)(P–P)(bipy)]PF6, where L = Lap or Law, P–P = 1,2′-bis(diphenylphosphino)ethane (dppe), 1,4′-bis(diphenylphosphino)butane (dppb), 1,1′-bis(diphenylphosphino)ferrocene (dppf) and bipy = 2,2′-bipyridine, were synthesized, fully characterized by elemental analysis, molar conductivity, NMR, cyclic voltammetry, UV-vis, IR spectroscopies and three of them by X-ray crystallography. All six complexes were active against breast (MCF-7 and MDA-MB-231) and prostate (DU-145) cancer cell lines with lower IC50 values than cisplatin. Complex [Ru(Lap)(dppe)(bipy)]PF6 (1a) showed significant selectivity for MDA-MB-231, a model of triple-negative breast cancer (TNBC), as compared to the “normal-like” human breast epithelial cell line, MCF-10A. Complex (1a) inhibited TNBC colony formation and induced loss of cellular adhesion. Furthermore, the complex (1a) induced mitochondrial dysfunction and generation of ROS, as is involved in the apoptotic cell death pathway. Preferential cellular uptake of complex (1a) was observed in MDA-MB-231 cells compared to MCF-10A cells, consistent with the observed selectivity for tumorigenic vs. non-tumorigenic cells. Taken together, these results indicate that ruthenium complexes containing lapachol and lawsone as ligands are promising candidates as chemotherapeutic agents.
- This article is part of the themed collection: New Talent: Americas


 Please wait while we load your content...
Please wait while we load your content...