Influence of magnetic dilution on relaxation processes in a solid solution comprising tetrahedral Co/ZnII complexes†
Abstract
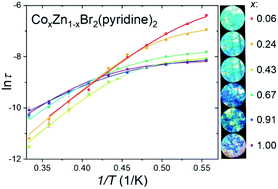
Single ion magnets have long been considered good prospective candidates to record a bit of information. One of the smallest known single ion magnets is CoBr2(pyridine)2. This molecular compound exhibits slow relaxation of magnetization mainly due to the thermally activated Orbach process, [A. M. Majcher et al., Chem. Sci., 2018, 9, 7277–7286]. However, the total relaxation time is dramatically shortened at low temperatures due to the direct, Raman, and quantum tunneling of magnetization processes. At low temperatures, the distribution of the probability of the possible relaxation pathways in this case favours QTM and the direct process over the Orbach process. To prolong the relaxation time, the compound was diluted with diamagnetic ZnII, producing 5 analogues of the general formula: CoxZn1−xBr2(pyridine)2 (x = 0.91, 0.67, 0.43, 0.24, and 0.06), confirmed to be a solid solution by independent experimental techniques (powder X-ray diffraction, infrared spectroscopy). The presence of diamagnetic ZnII ions changes the distribution of the dipolar interactions between the CoII centres in the material, which results in a monotonous change in the relaxation times, which in turn become longer with increasing dilutions, which is explained by the diminishing QTM contribution. The appearance of multiple relaxation processes is also observed for higher x, which is explained as the creation of multiple, separate frequency domains, as a result of the competition between QTM and the direct process contributions. We present a thorough, systematic study of magnetic dilution, which will hopefully be useful to estimate optimal dilutions in similar solid solutions.


 Please wait while we load your content...
Please wait while we load your content...