Modulation of the driving forces for adsorption on MIL-101 analogues by decoration with sulfonic acid functional groups: superior selective adsorption of hazardous anionic dyes†
Abstract
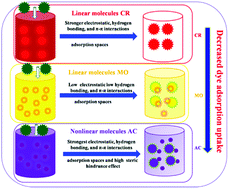
Toxic dyes pose a dramatic threat to human health and the environment when they are directly released into the water environment. Therefore, new porous materials with excellent adsorption performances are urgently needed to remove toxic dyes from wastewater. Herein, we report a sulfo-modified metal–organic framework (MIL-101-SO3H), which exhibits superior adsorption performance toward toxic dyes. The free –SO3H groups were successfully introduced into the MIL-101(Cr) skeleton to modulate the adsorption driving forces (i.e., hydrogen bonding, electrostatic attractions, and π–π stacking interactions together with adsorption spaces and steric hindrance) between the toxic dyes and MIL-101-SO3H adsorbent by varying the monosodium 2-sulfoterephthalic acid-to-terephthalic acid molar ratio in the reaction mixture. The experimental adsorption uptake by MIL-101-SO3H-1 was found to be 688.9 (methyl orange), 2592.7 (Congo red), and 213.2 (acid chrome blue K, AC) mg g−1, which was respectively 69.6, 89.6, and 51.5% higher than those of undecorated MIL-101. This adsorption behavior was attributed to the dye molecular structure, pore volumes, surface charge status, and functional groups of the adsorbents as well as the adsorption capacity of the dyes. The results revealed that the uncoordinated –SO3H groups increased the electrostatic attraction and hydrogen bonding between the MIL-101-SO3H adsorbent and linear anionic dyes. This increased the adsorption capacity toward the linear anionic dyes, overriding the effect of the pore volumes. On the other hand, the steric hindrance between the nonlinear anionic dye AC and MIL-101-SO3H adsorbent decreased the AC adsorption uptake of the latter, overriding the effects of hydrogen bonding and electrostatic attraction. In addition, the adsorption performance toward the cationic dye methylene blue increased, owing to the hydrogen bonding interactions, thereby overriding the effect of electrostatic repulsion and adsorption spaces. Thus, it is concluded that the adsorption behavior of the modified MIL-101-SO3H adsorbent is governed by the synergetic interplay between the electrostatic attraction, hydrogen bonding, and π–π stacking interactions as well as the steric hindrance and pore volumes of the adsorbent.


 Please wait while we load your content...
Please wait while we load your content...