Synthesis of Co2−xNixO2 (0 < x < 1.0) hexagonal nanostructures as efficient bifunctional electrocatalysts for overall water splitting†
Abstract
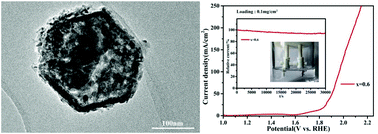
Designing low-cost and high-performance bifunctional electrocatalysts towards hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) is vitally important for water splitting. Herein, we synthesize Co2−xNixO2 (0 < x < 1.0) hexagonal nanosheets with different Co/Ni molar ratios via a facile coprecipitation process followed by calcination under an Ar atmosphere. Changing the Co/Ni molar ratios of the Co2−xNixO2 products is found to have a momentous influence on the microstructures, specific surface areas and electrocatalytic performances. At a Co/Ni molar ratio of 0.6, the Co1.4Ni0.6O2 nanosheet exhibits the largest specific surface area of 60.63 m2 g−1, the best OER with an onset overpotential of 278.5 mV, and HER of 72.8 mV as a bifunctional electrocatalyst. Meanwhile, the minimum Tafel slope is 113.6 mV dec−1 for OER and 77.4 mV dec−1 for HER. The Co1.4Ni0.6O2 nanosheet has excellent OER and HER activity at 0.1 mg cm−2 trace loading. Moreover, we construct an overall water splitting cell using the Co1.4Ni0.6O2 bifunctional electrocatalyst in a two-electrode system to further demonstrate the practical application, which needs a cell voltage of 1.75 V at a current density of 10 mA cm−2 and exhibits great long-term stability. These results provide an efficient strategy for the rational design of Co-based oxides towards bifunctional overall water electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...