Accelerating charge transfer at an ultrafine NiFe-LDHs/CB interface during the electrocatalyst activation process for water oxidation†
Abstract
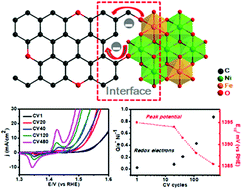
Owing to the combination of intriguing activity and conductivity, hybrid compositions of layered double hydroxides (LDHs) and carbon-based materials have been extensively and widely applied to evolve oxygen gas during water splitting. Here, a facile in situ nucleation strategy was used to construct ultrafine NiFe-LDH nanosheets monodispersed on a carbon black (CB) substrate. Notably, this work displayed the interfacial impact of combining CB with NiFe-LDHs on electrocatalyst activation. Interestingly, the optimized NiFe-LDHs/CB composite displays a fast activation rate and excellent water oxidation performance on a glassy-carbon electrode (an overpotential of 226 mV at 10 mA cm−2; a Tafel slope of 57 mV dec−1). This is due to the high active area, low impedance and ultra-high active metal atom utilization rate, accelerating charge transfer at the interface during the activation process. More importantly, this work highlights the interfacial charge transfer effect during the activation process and supplies clues for designing electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...