Temperature sensing performance based on up-conversion luminescence in hydrothermally synthesized Yb3+/Er3+ co-doped NaScF4 phosphors†
Abstract
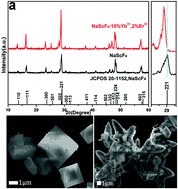
NaScF4:Yb3+/Er3+ crystals were successfully hydrothermally synthesized using distilled water as a single solvent. The crystal phase and morphology were characterized by XRD and SEM. Compared with organic solvents such as ethanol, ethylene glycol and ether, the molar ratio of Na/F/Sc plays an important role in the synthesis of NaScF4 crystals. Morphological control was achieved by changing the types of additives, and the relationship between morphology and luminescence properties was explored. The NaScF4:Yb3+/Er3+ phosphors exhibit strong green and red UC emissions under the excitation of 980 nm NIR. Optimal concentrations of Yb3+ and Er3+ for up-conversion luminescence performance were identified as 10% and 2%, respectively. Without any subsequent heat treatment process, the obtained NaScF4:10%Yb3+/2%Er3+ showed good temperature sensitivity. The temperature sensing ability was investigated by employing the dependence of the fluorescence intensity ratio (FIR) of the two thermal coupling energy levels of Er3+ (2H11/2 → 4I15/2 and 4S3/2 → 4I15/2) on temperature; the maximum sensitivity SA/SR was 0.00256 K−1/0.00317 K−1 at 548 K, and it increased to 0.00328 K−1/0.00413 K−1 after adding EDTA. In addition, an evaluation of temperature uncertainty during temperature measurement was performed, and was found to be 0.073 K and 0.095 K in the presence and absence of EDTA, respectively. Compared with some other reported materials, the obtained material shows a relatively superior temperature sensitivity, which provides new ideas for the improvement of temperature-sensitive materials.


 Please wait while we load your content...
Please wait while we load your content...