Uncovering lead formate crystallization in oil-based paintings†
Abstract
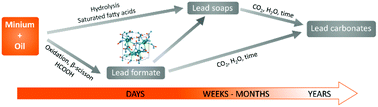
Lead carboxylates are an extensive group of compounds studied for their promising industrial applications and for their risky behavior when they are formed in oil paintings as corrosion products of lead-based pigments, leading to serious deterioration of paintings. Although the processes leading to the formation of aggregates, protrusions or inclusions, affecting undesirably the appearance of paintings, are assumed to be long term, neo-formed lead carboxylates are detectable in the early stage of paint drying. To uncover the chemical changes in lead pigments during the drying of oil paint films, model systems consisting of minium (Pb3O4) and four common drying oils were studied by X-ray powder diffraction (XRPD), 13C and 207Pb solid state NMR (ssNMR) spectroscopy and Fourier-transformed infrared spectroscopy (FTIR). For the first time, a degradation mechanism of Pb3O4via the crystallization of lead formate (Pb(HCOO)2), at the end of oxidative polymerization of oil paint films, was uncovered. The formation of formic acid in oils was proved by gas chromatography-mass spectrometry (GC-MS). Vapor experiments evidenced the susceptibility of Pb3O4 to react with volatile formic acid released during the autoxidation of oils comparably to the direct pigment−binder interactions in paint films. The investigation of the local environment of lead atoms in the paint film by 207Pb WURST-CPMG NMR spectroscopy showed that Pb(II) atoms reacted with linseed oil preferentially to form highly crystalline Pb(HCOO)2, while the local chemical environment of Pb(IV) atoms did not change. The results proved the co-existence of (i) highly crystalline Pb(HCOO)2, (ii) a highly mobile amorphous phase corresponding to free carboxylic acids or a nascent lead soap phase and (iii) the remaining Pb3O4 in the polymeric/ionomeric network. Pb(HCOO)2 is assumed to be an intermediate for the conversion of Pb3O4 to lead soaps and/or lead carbonates.


 Please wait while we load your content...
Please wait while we load your content...