Bulky bis(aryl)triazenides: just aspiring amidinates? A structural and spectroscopic study†
Abstract
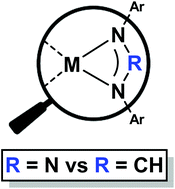
The synthesis and molecular structures (by single crystal X-ray diffraction) of s-, p- and d-metal complexes of the sterically demanding N,N′-bis(2,6-diisopropylphenyl)triazenide are reported and the spectroscopic (NMR spectroscopy and infrared spectroscopy) and physical properties of these complexes compared to related formamidinate complexes. Through the use of infrared spectroscopy the σ-donor capacity of this ligand is demonstrated to be reduced relative to the structurally isomorphous formamidinate congener, which supports previously advanced theoretical calcluations and DFT results reported herein. These electronic differences are highlighted by the stark contrast in reaction outcomes at rhodium; where [(Dipp2N3)Rh(CO)2] (1) is an isolable, stable complex and the formamidinate complex is not. The coordination chemistry of the triazenide ligand for the s-block metal complexes (M = Li, Na, K) has been shown to give structurally isomorphous complexes to the formamidinate analogue. In contrast to the amidinate complexes, these complexes show extreme lability of coordinated, volatile Lewis-bases, which in turn-yields the highly insoluble base-free triazenide complexes. These complexes are also synthesized directly in the absence of donor solvents. This triazenide ligand has proven to be a suitable ligand for stabilising reactive main group hydrides of Group 13 (M = Ga, In) and attempts at the analogous thallium hydride complex by halide–hydride exchange are reported. Finally attempts at the synthesis of low valent main group complexes are reported ([MIL], M = In, Ga) are also reported, which yield instead disproportionation products ([MIIIXL2], M = Ga, In; [{MIIXL}2], M = Ga).


 Please wait while we load your content...
Please wait while we load your content...