A novel binuclear Pd(ii) complex displaying synergic peptide cleavage behaviour†
Abstract
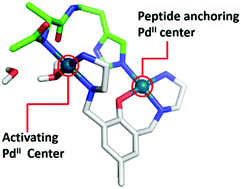
Mononuclear Pd(II) complexes with two leaving groups are able to promote His-, Cys- and Met-orientated peptide hydrolysis, and exploring the peptide cleavage behavior of a novel Pd(II) complex may provide “Omics” studies a promising artificial protease. In this study, a novel binuclear Pd(II) complex [Pd2(μ-O-L-H)(μ-Cl)](ClO4)2 (L = 2,6-bis(N-2′-aminoethylaminomethyl)-p-cresol) was constructed to promote peptide hydrolysis. Although each Pd(II) center has only one leaving group (Cl) in this complex, electrophoresis and LC-MS-MS determination discloses that this complex enables myoglobin cleavage on the second upstream peptide bond from His and Met. A study on peptide cleavage also confirms the His- and Met-orientated peptide hydrolysis, yet no Cys-orientated hydrolysis was observed, although the cysteine-induced peptide/complex binding is distinct. Cysteine in the peptide even prevents the complex from promoting His-orientated hydrolysis, whereas the oxidized cysteine residue recovers the His-orientated hydrolysis. This peptide cleavage behavior is quite different from the simultaneous His-, Cys-, and Met-orientated hydrolysis promoted by the mononuclear Pd(II) complexes. A theoretical study suggests that the two Pd(II) centers of this complex might promote His- and Met-orientated hydrolysis in a synergic manner: one Pd(II) center binds selectively on peptides or proteins and the other coordinates with the amide bond and water favoring nucleophilic attack on the peptide bond. The thiol group of cysteine is inclined to bridge the two Pd(II) centers to form a “closed” sulphur-bridged structure, disfavoring the Cys-orientated hydrolysis. This study not only demonstrates the peptide cleavage behavior of this binuclear Pd(II) complex, but also provides a polynuclear strategy to regulate the peptide cleavage behavior of Pd(II) complexes.


 Please wait while we load your content...
Please wait while we load your content...