Preorganized helical chirality controlled homochiral self-assembly and circularly polarized luminescence of a quadruple-stranded Eu2L4 helicate†
Abstract
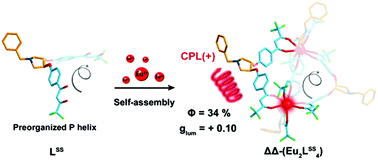
β-Diketones are one of the most widely used ligands for sensitizing the luminescence of lanthanide complexes due to their excellent sensitization abilities. However, the difficulties in introducing chiral groups to take part in the electronic transitions of conjugated systems limit their application in lanthanide circularly polarized luminescence (CPL) materials. In view of the inherent chirality of the helical structure, herein, a pair of homochiral quadruple-stranded helicates, Eu2L4, is assembled based on chiral bis-β-diketonate ligands, wherein the two point chirality centers in the spacer preorganize the helical conformation of the ligand (3S,4S)/(3R,4R)-3,4-bis(4,4′-bis(4,4,4-trifluoro-1,3-dioxobutyl)phenoxyl)-1-benzylpyrrolidine, LSS/LRR. X-ray crystallographic analyses reveal that the R,R configurations of the chiral carbons in the spacer induce the M helical sense of the ligand, while the S,S configurations induce the P helical sense. Through the comprehensive spectral characterization in combination with semiempirical geometry optimization using the Sparkle/RM1 model, it is confirmed that the preorganized ligands successfully control the homochirality of the helicates. Moreover, the mirror-image CD and CPL spectra and NMR measurements confirm the formation of enantiomeric pairs and their diastereopurities in solution. Detailed photophysical and chiroptical characterization studies reveal that the helicates not only exhibit intense circularly polarized luminescence (CPL) with |glum| values reaching 0.10, but also show a high luminescence quantum yield of 34%. This study effectively combines the helical chirality of the helicates with the excellent sensitization ability of the β-diketones, providing an effective strategy for the syntheses of chiral lanthanide CPL materials.


 Please wait while we load your content...
Please wait while we load your content...