Structure and thermal expansion behavior of Ca4La6−xNdx(SiO4)4(PO4)2O2 apatite for nuclear waste immobilization†
Abstract
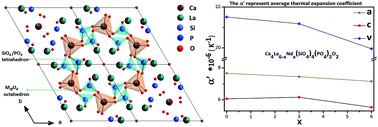
In this study, Ca4La6−xNdx(SiO4)4(PO4)2O2 (x = 0, 1, 2, 3, 4, 5, and 6) apatites were explored for nuclear waste immobilization, and Nd3+ ions were used as the surrogate of radionuclides (such as Am3+, Cm3+, and Pu3+). The synthesized samples conform to the P63/m (176) symmetry in the hexagonal system according to the characterizations by means of X-ray diffraction, Raman spectra, and Fourier-transform infrared spectra. Rietveld analyses indicate that both Ca2+ and Ln3+ (La, Nd) cations are located at the M4f and M6h sites, which is different from earlier studies. The M6h sites prefer to be occupied by Ln3+ (La, Nd) cations with higher valence. Besides, the content of the impurity phase Ca3(PO4)2 reduces from 2.815 wt% to 0 with the incorporation of Nd3+ ions. These results demonstrate that apatites possess excellent ability to accommodate radionuclides with various valences and radii at the M4f and M6h sites. Moreover, we investigated the thermal expansion behavior by high-temperature X-ray diffraction. There is no phase transformation in the range of 298–1173 K, and the Ca4La6−xNdx(SiO4)4(PO4)2O2 apatites exhibit lower thermal expansion coefficients than other candidates that have been extensively studied. Furthermore, the thermal expansion coefficient gradually decreases with the accommodation of Nd3+ ions. All the results suggest that apatites are promising candidates for nuclear waste immobilization.

 Please wait while we load your content...
Please wait while we load your content...