The development of two fluorescent chemosensors for the selective detection of Zn2+ and Al3+ ions in a quinoline platform by tuning the substituents in the receptor part: elucidation of the structures of the metal-bound chemosensors and biological studies†
Abstract
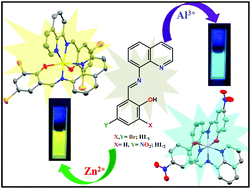
Here, two 8-aminoquinoline-based chemosensors, namely, HL1 and HL2 (HL1 = 2,4-dibromo-6-((quinolin-8-ylimino)methyl)phenol and HL2 = 4-nitro-2-((quinolin-8-ylimino)methyl)phenol) were synthesized by simply changing the substituents in the ligand framework under ambient conditions. They were thoroughly characterized using different spectroscopic techniques, including ESI-mass spectrometry and elemental analysis. HL1 selectively sensed Zn2+ ions, whereas HL2 detected Al3+ ions. The metal-bound chemosensors (complexes 1 and 2) were also investigated using different techniques including X-ray crystallography. The binding stoichiometry of the probes with the respective ions was confirmed to be 2 : 1 by Job's plot analysis and X-ray crystallography. The limit of detection (LOD) values for both chemosensors towards the respective metal ions were in the order of ∼10−7 M, which clearly indicates that these probes have significant potential for biological applications. The capability of our synthesized chemosensors to detect intracellular Zn2+ and Al3+ ions in the triple negative human breast cancer cell line MDA-MB-468 was evaluated with the aid of fluorescence imaging. Mechanistic insights into the anticancer activity of complexes 1 and 2 were also demonstrated in this study. To the best of our knowledge, this is the first time that this type of biological and sensing activity for 8-aminoquinoline-based complexes has been demonstrated in a single platform.


 Please wait while we load your content...
Please wait while we load your content...