Pristine S,N-containing Mn-based metal organic framework nanorods enable efficient oxygen reduction electrocatalysis†
Abstract
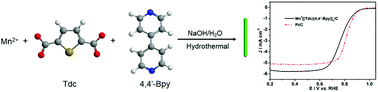
Owing to their unique physicochemical properties, metal–organic frameworks (MOFs) are a kind of promising material for electrocatalysis. However, many reports focus on the use of MOFs as precursors to produce efficient electrocatalysts by pyrolysis. The use of pristine MOFs with well-defined structures as efficient electrocatalysts directly is still a challenging problem. Herein, S,N-containing MnII[(Tdc)(4,4′-Bpy)]n with different morphologies have been obtained by using MnII, thiophene-2,5-dicarboxylate (Tdc) and 4,4′-bipyridine (4,4′-Bpy) as raw materials via hydrothermal synthesis. Furthermore, the influences of different hydrothermal reaction times (0, 2 and 4 h) and anions (SO42−, Cl−, NO3− and CH3COO−) on the morphologies, compositions and ORR activity of the resultant Mn-MOFs are also investigated at length. The results indicate that only the reaction of MnSO4 with Tdc and 4,4′-Bpy for 4 h can form relatively uniform one-dimensional (1D) MOF nanorods. The 1D nanorods combine the favorable features towards the oxygen reduction reaction (ORR), such as a high surface area and efficient 1D electron/mass transport capability. Therefore, the MnII[(Tdc)(4,4′-Bpy)]n nanorods display the highest ORR activity with an onset potential of 0.98 V and a half-wave potential of 0.78 V vs. RHE (reversible hydrogen electrode), which is even comparable to that of Pt/C. In addition, the MnII[(Tdc)(4,4′-Bpy)]n nanorods exhibit higher stability, methanol resistance and ORR selectivity than Pt/C. The present study illustrates an efficient fabrication strategy for highly efficient 1D MOF nanorods for energy storage and conversion applications.


 Please wait while we load your content...
Please wait while we load your content...