3D Ni and Co redox-active metal–organic frameworks based on ferrocenyl diphosphinate and 4,4′-bipyridine ligands as efficient electrocatalysts for the hydrogen evolution reaction†
Abstract
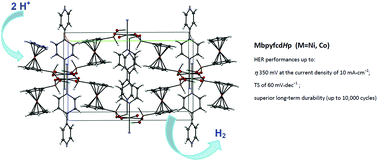
New 3D Ni and Co redox-active metal–organic frameworks based on ferrocenyl diphosphinate and 4,4′-bipyridine ligands have been synthesized, characterized by single crystal X-ray diffraction and spectroscopic techniques and explored as stable electrocatalysts capable of meeting two important parameters: the overpotential and Tafel slope (TS) in the hydrogen evolution reaction (HER). The electrochemical studies suggest that the reaction kinetics of a Ni-MOF (1) catalyst is more favorable than that of a Co-MOF (2) catalyst. Particularly, Ni-MOF exhibits better HER performance with an overpotential of 350 mV at a current density of 10 mA cm−2, a small TS of 60 mV dec−1 and superior long-term durability (of up to 10 000 cycles), ranking it among the most active non-noble metal-based molecular electrocatalysts. The introduction of a 4,4′-bpy linker in 2 significantly changes the catalytic properties in an organic or aqueous environment compared to 1D cobalt polymers based on ferrocenyl diphosphinate. For Co-MOF 2, there is a significant decrease in the overvoltage by ∼440 mV in comparison with the 1D Co polymer in an organic medium and by 50 mV in an aqueous medium. The TS changes from 120 to 65 mV dec−1 when moving from 1D CofcdHp to a 3D structure of 2. Thus, a 4,4′-bpy linker reduces the overvoltage and gives more favorable HER kinetics (lower TS). These results provide important guidelines for the rational design of non-precious metal electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...