Modulating energetic performance through decorating nitrogen-rich ligands in high-energy MOFs†
Abstract
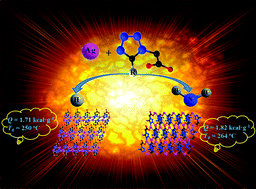
In the presence of different nitrogen-rich ligands, two energetic MOFs with formulas [Ag(tza)]n (1) and [Ag(atza)]n (2) (Htza = tetrazole-1-acetic acid and Hatza = (5-amino-1H-tetrazole-1-yl) acetic acid) were successfully synthesized and characterized. X-ray single crystal structure analysis shows that both 1 and 2 have 2D layer-like topologies. The experimental and theoretical evaluations reveal the promising properties of both energetic compounds, such as prominent heats of detonation, high thermal stabilities, good sensitivities and excellent detonation performances. In contrast to 1, interestingly, the introduction of the amino group in 2 leads to various coordination modes of the ligands and different stacking patterns of the frameworks, resulting in the observation of the shorter Ag–O, Ag–Ag, C–N, N–N, and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond lengths in 2. Consequently, 2 features superior heats of detonation and thermostability compared to 1. The nonisothermal thermokinetic parameters are obtained by using the Kissinger and Ozawa methods, while the standard molar enthalpies of formation are calculated from the determination of constant volume combustion energies. In addition, both compounds were explored as practical additives to promote the thermal decomposition of ammonium perchlorate (AP). This work may provide an effective approach for manipulating the energetic properties and thermostability of high-energy compounds via the perturbation of energetic groups.
N bond lengths in 2. Consequently, 2 features superior heats of detonation and thermostability compared to 1. The nonisothermal thermokinetic parameters are obtained by using the Kissinger and Ozawa methods, while the standard molar enthalpies of formation are calculated from the determination of constant volume combustion energies. In addition, both compounds were explored as practical additives to promote the thermal decomposition of ammonium perchlorate (AP). This work may provide an effective approach for manipulating the energetic properties and thermostability of high-energy compounds via the perturbation of energetic groups.


 Please wait while we load your content...
Please wait while we load your content...