Ligand-based electronic effects on the electrocatalytic hydrogen production by thiosemicarbazone nickel complexes†
Abstract
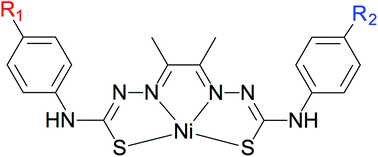
This work reports on the synthesis and characterization of a series of mononuclear thiosemicarbazone nickel complexes that display significant catalytic activity for hydrogen production in DMF using trifluoroacetic acid as the proton source. The ligand framework was chemically modified by varying the electron-donating abilities of the para substituents on the phenyl rings, which was expected to impact the capability of the resulting complexes to reduce protons into hydrogen. Over the four nickel complexes that were obtained, the one with the thiomethyl substituent, NiSCH3, was found to overtake the catalytic performances of the parent complex NiOCH3 featuring lower overpotential values and similar maximum turnover frequencies. These results confirm the electronic effects of the ligand on HER when using thiosemicarbazone nickel complexes and support that chemical modifications can tune the catalytic performances of such systems.
- This article is part of the themed collection: Inorganic Reaction Mechanisms


 Please wait while we load your content...
Please wait while we load your content...