Hybrid iodoplumbates with metal complexes: syntheses, crystal structures, band gaps and photoelectric properties†
Abstract
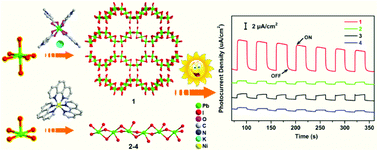
With the in situ-generated [Pb(MCP)4]2+ (HMCP+ = 1-methyl-4-(carboxyl)pyridinium) or [M(phen)3]2+ (M = Co, Fe and Ni; phen = 1,10-phenanthroline) complexes as structural directing agents and charge-balancing ions, we solvothermally synthesized and structurally characterized four new organic–inorganic hybrid iodoplumbates. Compound K2[Pb(MCP)4]Pb3I10 (1) represents the first K+ and [Pb(MCP)4]2+ co-templated hybrid haloplumbate, and exhibits a curve-like anionic layer of [Pb3I10]n4n−. Compounds [M(phen)3]Pb2I6·CH3CN (M = Co (2), Fe (3) and Ni (4)) have isostructural phases, and feature a one-dimensional (1D) [Pb2I6]n2n− anionic chain characteristic of pyramid-like [PbI5] units. The optical property studies show that compounds 1–4 exhibit semiconductor behaviors with the band gaps of 1.98–2.68 eV. In addition, the title compounds exhibited interesting photoelectrical responsive properties, with the photocurrent density in the order of 1 > 3 > 2 > 4. The thermal stabilities of the title compounds 1–4, as well as the theoretical band structure and density of states (DOS) of compounds 1 and 2 have also been studied.


 Please wait while we load your content...
Please wait while we load your content...