Mechanistic study of carboxylic acid and phosphate ester cleavage by oximate metal complexes surpassing the limiting reactivity of highly basic free oximate anions†
Abstract
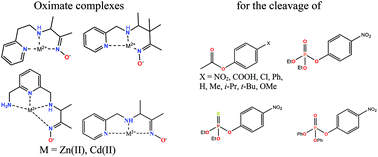
Two tridentate and one tetradentate new ligands containing the terminal oxime group separated from secondary amino and pyridine groups as additional binding sites by two or three methylene groups have been prepared. Their acid–base properties, as well as the composition and stability of their complexes with Zn(II) and Cd(II) ions, were determined by potentiometric and spectrophotometric titrations. The X-ray structure of a Cd(II) complex of a related tridentate oxime ligand previously studied in solution was determined. All oximate complexes show high reactivity in the cleavage of aryl acetates, paraoxon, parathion and 4-nitrophenyl diphenyl phosphate, with rate constants significantly surpassing the limiting rate constants observed for highly basic free oximate anions. The second-order rate constants for individual oximate complexes in solution are assigned to each ligand, metal cation and substrate. The results of the cleavage of 4-substituted phenyl acetates were analyzed in terms of Brønsted correlations with the leaving group pKa, which demonstrated a change in the rate determining step from the nucleophilic attack to the leaving group departure upon an increase in the leaving group basicity. The zero slope of the Brønsted correlation for the nucleophilic attack indicates transition state stabilization through electrophilic assistance by the metal ion. This interpretation is supported by metal selectivity in the relative efficiency of the cleavage of paraoxon and parathion. The existence of the alpha-effect in ester cleavage by coordinated oximates is confirmed by an analysis of the Brønsted correlations with the nucleophile basicity for metal bound oximate and alkoxo or hydroxo nucleophiles. The very high reactivity of the oximate complexes of the new ligands is attributed to transition state stabilization and to the removal of the solvational imbalance of oximate anions that impedes the expected increase in the reactivity of highly basic free anions.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...