Efficiency and mechanism of adsorption of low-concentration uranium from water by a new chitosan/aluminum sludge composite aerogel
Abstract
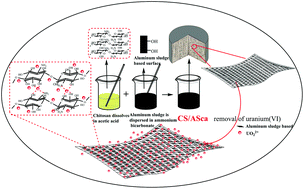
A new chitosan/aluminum sludge composite aerogel (CS/ASca) exhibiting good selectivity, easy separation potential, and high adsorption capacity was synthesized by combining chitosan (CS) and aluminum sludge from waterworks (AS). The adsorption of U(VI) by the CS/ASca was assessed as a function of solution pH, adsorption time, temperature, initial concentrations of uranium, and coexisting ions. The systematic batch experiments reveal that the adsorption kinetics is described by a pseudo-second-order model, and the sorption thermodynamics involves spontaneous endothermic processes. At a pH of 4, 308 K, and initial uranium concentrations of 10–700 mg L−1, the maximum adsorption capacity of the CS/ASca for U(VI) (simulated by the Langmuir model) was 434.64 mg g−1. Data from scanning electron microscopy/energy dispersive spectrometry, Fourier-transform infrared, and X-ray photoelectron spectroscopy indicated that uranyl ion adsorption was predominantly associated with the complexation of U(VI) with the amino and hydroxyl groups on the surface of the CS/ASca. In addition, our results demonstrated that the Mg(II), Pb(II), Na(I), and K(I) ions had little or no effect on the sorption of U(VI) on the CS/ASca. This study provides new clues for the treatment of radioactive wastewater.


 Please wait while we load your content...
Please wait while we load your content...