Designing of novel zinc(ii) Schiff base complexes having acyl hydrazone linkage: study of phosphatase and anti-cancer activities†
Abstract
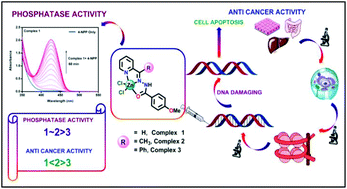
Three asymmetric tridentate acyl hydrazone Schiff base ligands namely L1, L2 and L3 were prepared via condensation of 4-methoxybenzohydrazide with picolinaldehyde, 1-(pyridin-2-yl)ethanone and phenyl(pyridin-2-yl)methanone respectively. Three bio-relevant mononuclear zinc(II) complexes [Zn(L1)Cl2]·2H2O (1), [Zn(L2)Cl2] (2) and [Zn(L3)Cl2] (3) were synthesized by treatment of zinc(II) chloride with the corresponding Schiff base ligands and characterised by the usual physicochemical techniques. The solid state structures of complexes 1 and 3 were evaluated by single crystal X-ray analysis. All complexes were able to hydrolyse the P–O bond of the phosphate monoester in 90% (v/v) DMSO–water medium using 4-nitrophenylphosphate (4-NPP) as the model substrate and the trend in their activity is 1 ≈ 2 > 3. On considering the highly efficient hydrolysis properties, complexes 1–3 were tested as potential therapeutic agents for cancer using HCT116 (human colorectal carcinoma), HepG2 (human hepatocellular carcinoma) and A549 (human non-small lung carcinoma) cells. Complex 2 showed the highest IC50 values for anti-cancer activity towards all three cell lines among the three complexes and complex 3 showed the least activity as observed in the phosphatase activity study. The internucleosomal degradation of DNA during apoptosis can be detected by cell death detection ELISA. DNA fragmentation that leads to cell death was examined and when induced by complex 2 in HepG2 cells a significant level of DNA fragmentation was observed at regular intervals of time.


 Please wait while we load your content...
Please wait while we load your content...