Direct hydrothermal synthesis of amine-functionalized cubic hematite (C-Fe2O3) and sonochemical deposition of nanosized Au for its application as a visible-light photocatalyst†
Abstract
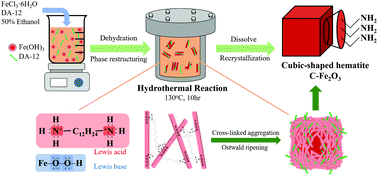
Cubic-shaped hematite (C-Fe2O3) functionalized with amine groups was directly prepared via one-pot hydrothermal reaction of Fe3+ with 1,12-diaminododecane (DA-12) in aqueous solution (50% ethanol). Herein, DA-12 (as a Lewis acid) promoted the aggregation of α-FeOOH nanorods with Lewis base sites, leading to the rapid recrystallization and conversion into uniform C-Fe2O3. C-Fe2O3 was subsequently deposited with nanosized Au via sonochemical reduction of 1.0 wt% HAuCl4 (0.1–0.8 mL), hereafter referred to as Au-deposited C-Fe2O3 (C-Fe2O3@Au). X-ray diffraction patterns of C-Fe2O3@Au confirmed the hexagonal crystalline phases of hematite and crystalline Au (111) and showed a weak broad band attributed to the amorphous carbon of DA-12. C-Fe2O3@Au was tested as a visible-light photocatalyst towards the degradation of methylene blue (MB) dye. C-Fe2O3@Au (0.1–0.4 mL of 1.0 wt% HAuCl4) exhibited 6–8 times higher photocatalytic activity than the Au-free counterpart (C-Fe2O3). The enhanced photocatalysis was mainly attributed to the improved separation efficiency of photo-excited charge carriers, i.e., the facilitated transport of electrons from the conduction band to the lower lying Fermi level of Au. However, the photocatalytic activity of C-Fe2O3@Au (0.8 mL of 1.0 wt% HAuCl4) was decreased probably due to the reduction of active sites for MB adsorption by the high coverage of the Au layer. The combined hydrothermal and sonochemical methods provided the direct synthetic route to cubic-shaped hematite decorated with nanosized Au and surface amine functionality as a promising visible-light photocatalyst.


 Please wait while we load your content...
Please wait while we load your content...